Buffers and solutions
Buffer Solutions – pH Stabilization in Laboratory Analysis
Buffer solutions are an essential part of the modern laboratory, especially where buffer pH control is critical to chemical and biological reactions. Each buffer solution makes it possible to maintain a relatively constant pH even with the addition of small amounts of acids, bases or during dilution.
As a result, pH buffers are widely used in chemical analysis, pharmaceuticals, biotechnology, and in industrial processes related to quality control and substance property testing.
Physicochemical basis of buffer systems
The basic definition of a buffer solution is based on the presence of a conjugate pair: a weak acid and its salt, or a weak base and its salt. The classic definition of a buffer solution indicates that such a system counteracts pH changes through neutralization reactions.
For example:
- Acid buffer: CH₃COOH + CH₃COONa
- alkaline buffer: NH₃ + NH₄Cl
Reactions responsible for stabilizing the pH of the buffer solution:
- neutralization of H⁺ ions: CH₃COO⁻ + H⁺ → CH₃COOH
- neutralization of OH⁻ ions: CH₃COOH + OH⁻ → CH₃COO⁻ + H₂O
A quantitative description is provided by the Henderson-Hasselbalch equation: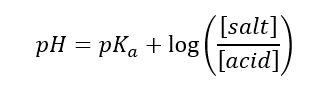
It is these relationships that allow you to precisely determine the pH of the buffer and its range of action.
The role of buffer systems in physiology and pharmacology
pH buffer solutions play an important role not only in chemistry, but also in biological systems. In living organisms, they are responsible for maintaining a stable environment of enzymatic reactions, where even a small change in the pH of the buffer solution can disrupt the functioning of proteins.
In pharmacy, pH buffers are used in drug formulations to ensure their shelf life and safety. In the food and cosmetics industries, buffer solutions stabilize the properties of the end products.
Optimization and stabilization of parameters in laboratory conditions
In laboratory practice, the preparation of an appropriate buffer solution requires precision and control of parameters. Use tools such as laboratory meters, including portable pH meters and benchtop pH meters, that allow you to accurately measure the pH of the buffer. In more advanced analyses, oxygen meters are also used.
Buffer Solution Preparation Process:
- selection of the buffer system (preferably using a buffer calculator)
- dissolving ingredients in about 80-90% of the target volume of water
- Thorough mixing and pH measurement
- pH correction using 0.1–1 M HCl or NaOH
- Filling the volume in the volumetric flask to the final value
Buffer solutions prepared in this way ensure stability and repeatability of results in laboratory analyses.
Specificity and operating ranges of selected analytical mixtures
The selection of the appropriate pH buffers depends on the required range of action. Different pH buffer solutions work efficiently in specific ranges:
- pH 2-3: phthalate buffer
- pH 4-5: acetate buffer (CH₃COOH + CH₃COONa)
- pH 6–8: phosphate buffer (NaH₂PO₄ / Na₂HPO₄ – hydrosol system)
- pH 7-8: HEPES (biochemistry)
- pH 8–9: TAE (Tris-acetate-EDTA)
- pH 9–10: borate buffer
- pH >10: carbonate buffer
Each such buffer solution has a specific buffer capacity and range of action, usually ±1 pH unit relative to pKa.
With the proper selection and preparation of buffer solutions, it is possible to maintain stable reaction conditions, which is crucial in modern property studies and chemical and biological analyses.






